Photocatalysis and Photocatalysts
The acceleration (increase in the reaction rate) of a photo reaction due to a photocatalyst, which is itself not consumed during the reaction, is called photocatalysis. The photcatalyst utilizes the energy of light (UV-VIS). Typically photocatalytic materials are based on titanium dioxide. Titanium dioxide coated iron oxides represent a novel group of inorganic pigments bearing photocatalytic activity.
Photocatalytic titanium dioxide (TiO2)
- Pure titanium dioxide is a semiconductor and photocatalyst.
- In case of pigmentary TiO2, the most important white opaque pigment, the photoactivity is suppressed by high rutile content, crystal lattice doping, crystal size adjustment and surface treatments.
- To maximize photoactivity TiO2-based products have mostly anatase crystal structure, very small crystal size and no stabilizing inorganic surface treatment.
Photocatalysis - the mechanism
- Anatase and rutile titanium dioxide crystals have band gaps of 3.2 eV and 3.0 eV, respectively. Thus they can be activated by light with wavelengths below 388 nm and 410 nm.
- If anatase is irradiated by light with energy larger than its band gap, the electrons are lifted into the conduction band and at the same time electron holes are created.
- Depending on the circumstances the electron hole pairs can react with their surroundings (wanted) or simply recombine (unwanted). Regarding a reaction with their surroundings (moisture and oxygen), the electrons act as reducing agents (reduction O2 --> O2·–), whereas the holes can participate in oxidation reactions (oxidation OH– --> ·OH). The Resulting reactive radicals will then decompose many organic substances (e.g. VOCs), air pollutants or other compounds to water and CO2.
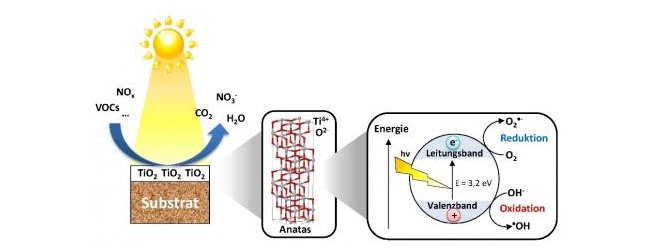
Figure: Activation of anatase-TiO2 and principle of photocatalysis
- To make activation by visible light possible, the band gap needs to be reduced. It can be done by introducing structural imperfections into the crystal. This is possible by varying the synthesis conditions and/or adding controlled amounts of impurities such as C, N, Fe3+, Cr3+ and others.
continue to Material testing

